Content
A recent meta-analysis published in Academic Radiology highlights the significant advancements in lymph node metastasis detection in colorectal cancer using MRI-based deep learning techniques. The study aggregated data from ten different studies, analyzing a total of 2,132 patients with colorectal cancer. The primary focus was to compare the diagnostic performance of deep learning models applied to MRI against that of unassisted radiologist interpretation. The results demonstrated that deep learning offered a 24 percent higher sensitivity than radiologists, showcasing its potential in improving detection accuracy.
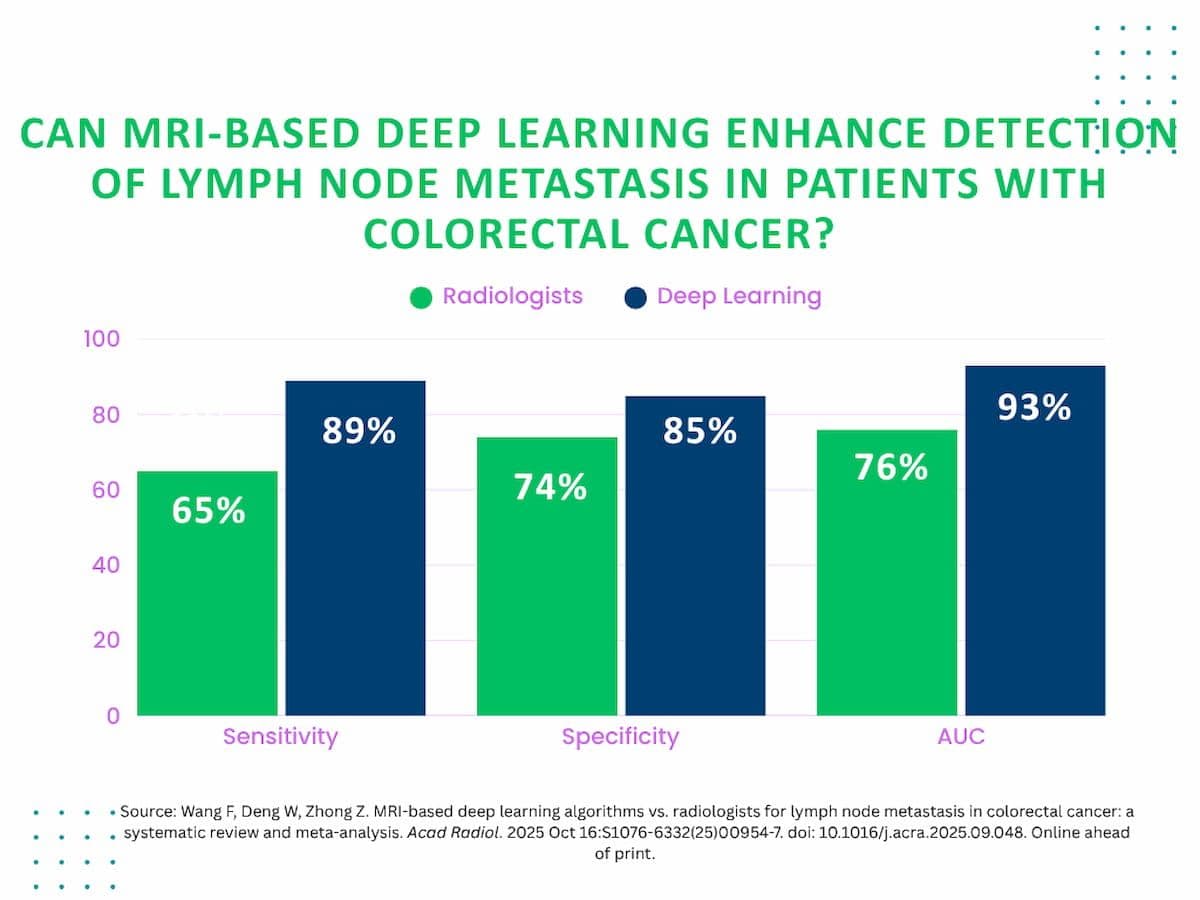
Within the internal validation cohorts, deep learning models achieved impressive pooled metrics, including a sensitivity of 89 percent, specificity of 85 percent, and an area under the curve (AUC) of 93 percent. In contrast, unassisted radiologist readings resulted in lower sensitivity at 65 percent, specificity at 74 percent, and an AUC of 76 percent. These figures underscore the superior diagnostic capabilities of deep learning systems when applied to colorectal cancer lymph node metastasis detection. External validation testing also revealed strong performance, with deep learning models maintaining a sensitivity of 75 percent, specificity of 81 percent, and an AUC of 84 percent.
The authors of the meta-analysis emphasize that MRI-based deep learning could serve as an effective adjunctive tool in clinical workflows. Such systems could function as first-pass readers, prioritizing suspicious cases and generating preliminary reports that radiologists can review and confirm. This approach may streamline workflow efficiency and reduce the time to diagnosis. Alternatively, deep learning models might act as concurrent readers, aiding radiologists in real time by highlighting regions of concern during image interpretation.
A notable finding from the analysis is the comparison between deep learning and radiologists of varying experience levels. While deep learning significantly outperformed unassisted radiologists, particularly junior ones, in terms of sensitivity and AUC, it did not show a statistically significant advantage in specificity when compared to senior radiologists. This suggests that experienced radiologists' ability to recognize complex benign imaging features remains a critical component of accurate diagnosis, and that deep learning tools are best positioned as complementary aids rather than replacements.
The study authors also discuss certain limitations within the meta-analysis. The majority of the included studies were retrospective and often limited to rectal cancer patients, with most cohorts composed primarily of Chinese individuals. These factors may restrict the generalizability of the findings to broader, more diverse populations. Additionally, the meta-analysis focused on the highest performing deep learning models from each study, which may not represent all current AI applications in this domain.
Overall, the findings support the integration of MRI-based deep learning into clinical practice for colorectal cancer staging, particularly for lymph node metastasis detection. The enhanced sensitivity and diagnostic accuracy of AI models could improve patient outcomes by facilitating earlier and more reliable identification of metastatic disease. Furthermore, the potential for workflow optimization through triage or concurrent reading strategies offers additional clinical value. Future research with more diverse and prospective cohorts will be essential to validate and expand upon these promising results.